














| Still deciding? Get samples of $ ! US$ 20/Piece Order Sample |
| Model NO. | Y3225 |
|---|---|
| Condition | New |
| Certification | ISO9001, ISO13485 |
| Standard | GB |
| Material | Stainless Steel (304, 316L) |
| Application | Medical Device Components, Metal Processing |
| Processing | Customized According to Drawings |
| Appearance | Electric Polishing |
| Process | Reducing, Expanding, Laser Cutting |
| Transport Package | Vacuum Packaging Carton |
| Specification | 3.0*160 |
| Production Capacity | 51,000 pieces/Year |
| Package Size | 30.00cm * 25.00cm * 15.00cm |
|---|---|
| Package Gross Weight | 3.800kg |

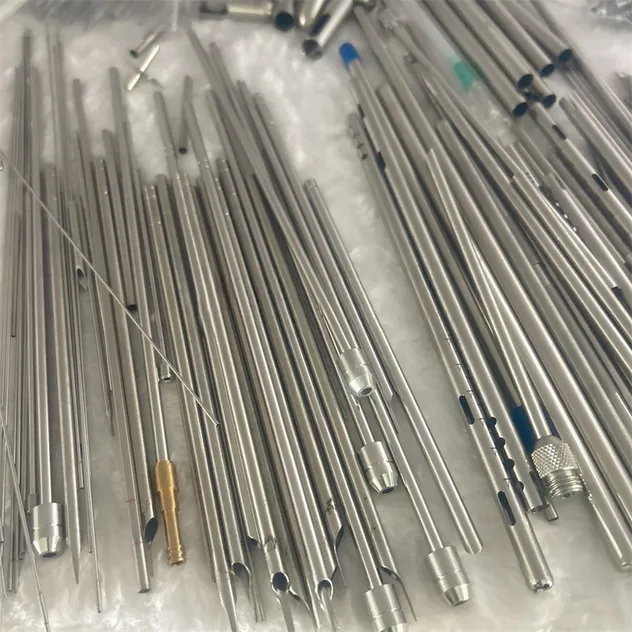

| Technical Specifications | Details | |
|---|---|---|
| Base Material | Material Grades | AISI 304, AISI 316L Stainless Steel |
| Properties | Corrosion resistance, high strength, biocompatibility | |
| Primary Processing | Laser Cutting | High-precision laser systems for clean cuts and micro-machining. |
| Tolerances | Dimensional | ±0.05mm or better depending on specifications. |
| Finishing | Sandblasting | Available in various grits for matte or smooth textures. |
| Forming | Flaring/Reducing | Precision expansion or diameter reduction of sections. |
| OD Range | Outer Diameter | Customizable from below 0.5mm upwards. |
| Wall Thickness | Thin-walled | Customizable from 0.1mm. |









A1: We primarily use medical-grade AISI 304 and AISI 316L stainless steel, known for their excellent corrosion resistance and biocompatibility.
A2: Yes, we provide full customization for Outer Diameter (OD), Inner Diameter (ID), wall thickness, and length based on your specific engineering drawings.
A3: Our capabilities include laser cutting, micro-grooving, sandblasting, flaring, diameter reduction, and electric polishing.
A4: Absolutely. We hold ISO 13485:2016 certification, which specifically governs quality management systems for medical devices.
A5: We typically achieve a dimensional tolerance of ±0.05mm or better, depending on the wall thickness and feature size.
A6: Yes, we provide both non-standard part prototyping and large-scale batch production processing.
 Flowtera Pipe
Flowtera Pipe